Written by Mike Hamilton, CCA & President of Turf Dietitian
Environmental pressure and golfers’ expectations have fostered the need for superintendents to comprehensively understand Nitrogen’s function within the plant and its precise management.
I know most Superintendents use fertilizers and chemicals responsibly, but many homeowners and less qualified individuals aren’t. Unfortunately, the world’s waters get environmentally impacted by others’ lack of experience applying Nitrogen. That impact should be of great concern to you.
If some environmental groups had their way, they would ultimately prohibit the use of Nitrogen on golf courses. You can bet that some environmental groups have already approached our legislators with such demands. The industry doesn’t need the fate of nitrogen fertilization used as a negotiating tool by legislators.

I’m not here to argue whether or not Nitrogen is polluting the Earth’s water, as that is above my level of expertise. What I am here to advocate for is the need to improve your knowledge continuously and to encourage you to make it your mission to use all fertilizers and chemicals as responsibly and efficiently as possible. Superior expertise will shield the golf industry against allegations of improper Nitrogen use.
The increasing demands from our Golfers have led to the need for precise nitrogen management. Golfers continue to ask for faster, smoother, and more playable greens. Unfortunately, job security gets based on your ability to provide those conditions without damaging your plant’s health. As we all know, that’s not an easy goal. The only way to keep the turf healthy and give the golfers what they want is through precision and effective management programs.
Low nitrogen levels can cause poor root growth, which in turn will cause insignificant uptake of water and nutrients. Lack of Nitrogen can also lead to poor color, thinning turf, poor disease resistance, and intensified environmental stress. Put more simply: if you under-apply Nitrogen, your turf will become sickly and probably die.
Over-application of Nitrogen to manicured turf can also cause life-threatening disease, significant deficiencies of other nutrients, environmental stress, and sleepless nights for Superintendents. Therefore, Nitrogen must be managed accurately. I’ve always believed no matter how good a person is at their job or how much of an expert they are. There is always room for improvement. So how do you get better at managing nitrogen usage?
Here is what you must know to ensure you’re doing your best:
- Monitor the plant’s need for Nitrogen at all stages of growth.
- Monitor nitrogen uptake and ingestion in the plant.
- Monitor plant health concerning nitrogen levels in the plant during all growth stages.
- Monitor nitrogen’s effect on the uptake of other nutrients.
- Monitor nitrogen loss from the soil.
- Monitor nitrogen loss from the plant.
- Monitor nitrogen levels in the soil.
- Monitor nitrogen levels in the irrigation source.
- Determine how much Nitrogen is released from soil organic matter.
- Determine the ideal nitrogen level in the plant, and only apply when levels are less than perfect.
- Have a complete understanding of the functions of Nitrogen in a plant.
- Apply the most efficient source of Nitrogen.
- Apply Nitrogen at the lowest rate necessary.
- Monitor weather conditions before scheduling any nitrogen application.
That all makes sense, right? So, how do you determine when to apply Nitrogen? That’s simple! Conduct water, soil, and plant tissue testing, along with the translation of all that data. By understanding where the N comes from, the levels of N in water and soil available for plant consumption, what source of N is present in the soil, how much N is in the plant, and how healthy the plant is, you will help to increase your expertise and efficiency.
Now the real question is, do you have the time to sift through all the data? To determine exactly when and how much Nitrogen to apply? Probably not. If you’re like most Superintendents, your plate is already full. However, that’s what Turf Dietitian is here for – that’s our specialty! We aim to be part of your team and assist you in making the most efficient and effective decisions for your nutrient management programs.
Choosing the Most Efficient Sources of Nitrogen
The application of the correct source is a critical component of any efficient nitrogen management program. Applying the exact source of Nitrogen for every application because it’s worked in the past can be wasteful. It can cause imbalances in other plant nutrients, diminishing plant health.
Mineralization, volatilization, nitrification, and denitrification are all factors that frequently change the form of Nitrogen. The weather also enormously impacts the fate and the transformation of Nitrogen in the soil. Unless you have a crystal ball, the only way I know to identify the amount and structure of Nitrogen in the soil is to have soil reserve, and soil solution tested. These tests should be standard practice for you and done at least four times per year in the south and three times per year in the north. (Spring, Summer, Fall, and Winter for warm season grasses).
You’ve all seen the chart that shows if a nutrient is a cation or anion. You also know that Nitrogen is both. N also gets taken up into the plant as an anion (NO3-) and cation (NH4+). Proper nitrogen management is dependent on the NO3 to NH4 ratio and total nutrient management.
Why is the ratio between NO3 and NH4 so important to nutrient management and plant health? Because even in this day, crowded with designer nutritional products, the most effective way to carry nutrients into the plant is with Nitrogen. Cations attach to NO3, and anions attach to NH4 because opposite charges attract. Given that the plant requires large amounts of Nitrogen, it will collect all it can through the mass flow of nutrients in the soil solution. When the Nitrogen goes into the plant, the attached nutrient goes along.
In reading many articles on this subject, the best I can determine is that the ideal NO3 to NH4 ratio in turf is 3 to 1. Why? Because cations are used by the plant in a higher quantity (about 3 to 1) to complete all its metabolic functions. If NH4 is high in soil solution, the plant will likely not take in enough cations. Conversely, if the NO3 is high in solution, the plant most likely will not take in enough anions.

Nitrogen (N) is critical for growing and sustaining the turf and plants on your property. This essential nutrient is the core element of most plant structures and the internal and external metabolic processes. So, what does this mean for your golf course? The turf on your golf course can’t live without Nitrogen.
Plants have one purpose on this Earth, to produce proteins and oxygen so that animal life to exist. Nitrogen is responsible for almost every process that converts light energy into plant proteins.
Known as the backbone for plants, Nitrogen ensures that your plants experience strong and healthy growth. Nitrogen is the nutrient used most by all plants and is essential for almost every metabolic and plant defense function. Given its significant influence on your plant’s health, it’s likely to apply Nitrogen inaccurately. Thus, Superintendents must monitor the nitrogen content in their soil and plant to ensure the plant is getting what it needs at all times.
The wellness of your plants depends on the availability and precise use of Nitrogen. Understanding the basic process of nitrogen behavior in soil may be complex, but it will make your life as a Superintendent much easier.

This biologically influenced process is, in turn, affected by climatic conditions. The location and soil composition of your golf course have an enormous influence on the fundamental biology of the system. The climate will also influence how Nitrogen behaves within the soil. You must get to know your soil to anticipate climatic changes.
Trust me, the more articles I read and learn about plant physiology, the more I am amazed by plants’ cellular development and their complexity. Each plant’s molecular structure needs nurturing. However, most of the customers I’ve dealt with have difficulty getting into the nitty-gritty science behind plant growth.
Think of it like this: your turf is a living organism, just like you. As you need food and water to survive, so does your turf. Knowing your plants and their needs will help you keep a healthy turf.
Nitrogen’s Primary Roles within the Plant:
To start with, let’s look at the role nitrogen plays in your plant’s structures. Nitrogen is a crucial nutrient for the health and development of your plants. A few reasons for this are:
Nitrogen makes up the plant structure’s amino acids – these are the building blocks of plant proteins. Plant proteins ensure healthy germination and the development of vital plant tissues and cells like the cell membranes and chlorophyll.
- Nitrogen aids the plant’s survival and helps form the plant’s DNA material.
- Nitrogen aids with photosynthesis and enhances the plant’s color.
- Nitrogen is vital for healthy growth and development.
- Nitrogen is a component of amino acids, proteins, vitamins, and enzymes.
Where Nitrogen Can Be Found:
We all know you can get bags of manufactured Nitrogen from your local fertilizer distributors. However, many times we ignore the most basic supplies. You must utilize the right amounts and embrace the natural nitrogen reserve in your soil.
On many occasions, there is enough Nitrogen released from soil organic matter during the growing season to supply much of the Nitrogen that the plant needs. Yet, most fertilizer suppliers developed programs that encourage applications of liquid forms of nitrogen weekly. But how does a supplier know your plant needs Nitrogen every week of the growing season? There is no possible way! Unless they are conducting a detailed analysis of every factor involved in the plant’s uptake and usage of nitrogen weekly.
Natural Nitrogen Sources:
Manures and Organic Waste: I’ve found that opinions on using natural organic fertilizers in the golf industry is usually divided down the middle. There seem to be two basic points of view on whether organic fertilizers are used or not used by Superintendents. Some Superintendents believe they are trying to prevent and reduce organic matter in their soils, so applying organic material will only add to the problem. Other Superintendents believe that organic fertilizers increase microbial activity in the soil, which helps to improve soil structure and reduces plant stress.
I believe the only bad thing about using organic sources is that the release of nutrients from the source is unpredictable. However, that is true with all slow-release sources. The reality is that no matter which philosophy you support, frequent monitoring is the only way to determine the plant’s release and usage of Nitrogen.
Clipping residues: Another source of Nitrogen is in clipping residues. Although very few golf courses return clipping on greens, I recommend doing it whenever possible. Despite existing in relatively small amounts, the clippings will help nourish both your turf and soil.
Clipping removal is not the only way that you may be disrupting the nitrogen flow in your golf course’s soil. Soil migration and runoff play a role, too. These may not be the main contributors to decreased nitrogen levels, but they should be considered when planning your water surface management.
I’d recommend developing a fertilizer program to help protect your course against nitrogen loss through erosion or runoff. Typically, the best programs I see combine bulk applications of slow-release products during aerification and liquids to supplement when needed.
Soil organic matter: We’d recommend developing a fertilizer program to help protect your course against nitrogen loss through erosion or runoff. Typically, the best programs I see combine bulk applications of slow-release products during aerification and liquids to supplement when needed.
By supplying oxygen to the soil, organic matter decomposes quickly. However, it leaves behind a more resistant residue that we call humus. This portion of organic matter releases about 20 to 40 pounds of Nitrogen per acre per year for each percentage of organic matter. It’s a much slower and more constant release of Nitrogen into the soil.
Other inputs of N for plant growth include:
- Atmosphere
- Biological fixation
- Atmospheric fixation
- Precipitation
- Commercial fertilizers
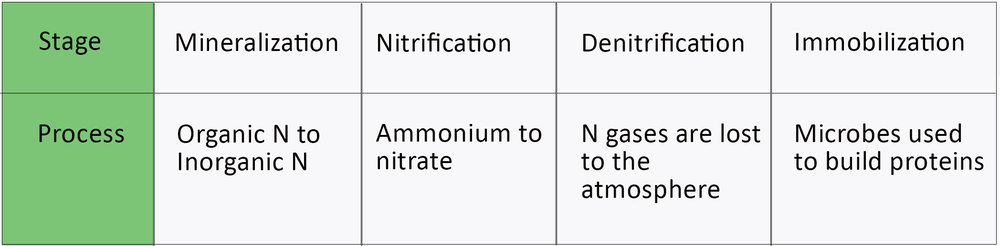
Nitrogen Transformations
As we’ve discussed, Nitrogen is known to change or transform its behaviors quite quickly. These changes influence the availability of Nitrogen for the plants.
Mineralization:
The conversion of organic to inorganic Nitrogen in soil’s OM, crop residues, and manure is called mineralization. Through this process, bacteria get digested in the organic material, and ammonium is released.
Nitrification:
Or the conversion of ammonium into nitrate. Nitrification proceeds rapidly in warmer, more moist soils that are well aerated.
Denitrification:
It is the process by which the converted bacteria that is now N gas will disappear into the atmosphere. Denitrification will occur in soil that is ample in organic matter and waterlogged. It should be noted that this process is generally kept to topsoil.
Immobilization:
The process happens when nitrate and/or ammonium are present in the soil, and the growing microbes use it to build proteins. Think of it as a shot of steroids that helps you grow to be big and strong.
During immobilization, the growing season has a net gain of N. And this happens due to the additional N present in the residue after the immobilization-mineralization processes.
Once you understand this cycle, you will be better equipped to guestimate what your grounds need during the different stages.
Your Nitrogen Cycle Cheat Sheet
Nitrogen loss from the soil system
To fully develop a plan of action to prevent Nitrogen loss, we have to back up and look at the cause. The loss of nitrate is a physical event referred to as leaching. This process involves the loss of NO3–N when it moves through the soil with excess water below the root zone. Simply, knowing your soil could make or break your soil system.
Each golf course has different types of soils with other functions. Coarse-textured soils have a lower water-holding capacity, with a higher potential to lose nitrates.
Various soil types also have different water-holding capabilities and affect how irrigation gets to your soil. Nitrogen can be filtered out of your soil if irrigation moves water straight through the root zone. If it moves past your root zone, your plant can’t benefit from nitrogen nutrients.
Denitrification can cause a loss in NO3–N when water saturates the soil for two or three days. Superintendents can take comfort in knowing that Nitrogen in the NH4+-N form isn’t subject to this loss.
Volatilization, on the other hand, can cause significant losses too. Volatilization occurs when Nitrogen escapes as ammonia gas. Levels of pH cause the release of ammonia gas. Urea can be found in manure and fertilizer and is the stimulant for the ammonia gas process. Incorporating Nitrogen into your manure and fertilizer will help eliminate the volatilization loss.
Nitrogen loss from volatilization is greater when:
- Levels of pH in the soil are higher than 7.3
- There’s a higher air temperature
- Moisture is rich on the soil’s surface
- The soil is rich in residue
To recap, here are the main factors contributing to nitrogen loss in the soil system:
- Leaching
- Denitrification
- Volatilization
- Crop Removal
- Soil erosion and runoff
- Improper amount of N applied
- Carbon-nitrogen ratios greater than 20:1
Biological processes influence the nitrogen cycle, which fluctuates with prevailing climatic conditions, and a particular soil’s physical and chemical properties. Both climate and soils can vary significantly across your golf course and affect N transformations for the different areas.
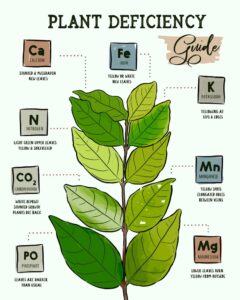
Nitrogen Deficiency Symptoms

Now that we know the causes, we can start identifying when your plants are taking a turn for the worst.
- The plants’ petioles and veins turn a light red.
- Older leaves will gradually change from a normal, healthy green appearance to a much paler one. They will become more uniformly yellow as the deficiency progresses.
- If your leaves turn yellow-white, they might have an extreme deficiency.
- Younger leaves tend to become smaller and have a paler green color.
- Your plants are shorter due to a reduced branching effect.
- The leaf’s veins will turn yellow along with the whole leaf’s appearance.
- A rosy-reddish or purple color shows on the underside of leaves and petioles.
- You will notice that older leaves will wilt under water stress much sooner than before the deficiency progressed.
Luckily, the recovery of deficient plants to applied nitrogen is immediate and spectacular.
Nitrogen Antagonism and Interaction
- Levels of micronutrients can become deficient when there are high levels of Nitrogen. Accelerated growth rates cause this.
- An excess of Nitrogen dilutes the Phosphorus, Calcium, Boron, Iron, and Zinc elements.
- Low soil levels can reduce the uptake of Phosphorus, Calcium, Boron, Iron, and Zinc.
- To make Molybdenum deficiency appear less noticeable, you can use Ammonium Nitrogen.
Key points for Superintendents
I hope for extensive transformations and reactions of Nitrogen in the soil. Were you expecting it to be so complex? Keep it simple and focus on these major points:
- Plants only take to inorganic Nitrogen, even though you can add both organic and inorganic nitrogen forms to your soil.
- All nitrogen sources can convert to NO3-N.
- Identifying the initial nitrogen source is impossible once it’s in the plant or water supply.
- Nitrate will move with your soil’s water and will always be present in the soil solution.
- Your plant uptake can increase due to inhibiting the conversion of NH4+-N to NO3-N. Less Nitrogen can be lost too. As a superintendent, you should keep sound management practices in place to help prevent the NO3-N from getting into the water supplies.
- You must test soil, water, and plant tissue as frequently as possible to precisely manage the plant’s nitrogen needs.
Common Nitrogen Sources
- Ammonium Nitrate
- Ammonium Sulfate
- Ammonium Polyphosphate
- Calcium Nitrate
- Potassium Nitrate
- Magnesium Nitrate
- Manganese Nitrate
- Trizone Urea
- Urea
- Sulfur Coated Urea
- Ureaformaldhyde
- IBDU
- Methylene ureas
- Organic Nitrogen
- 30-0-0
- 21-0-0—-24% Sulfur
- 10-34-0
- 25-0-0—-19% Calcium—-1.5% Magnesium
- 14-0-44
- 11-0-0—16% Magnesium
- 8-0-0—15% Manganese
- 46-0-0
- 36-0-0—16% Sulfur
- 36-0-0
- 31-0-0
- 38-0-0
- 3% to 10% N