Written by Mike Hamilton, CCA & President of Turf Dietitian

Because of that century and a half old Liebig’s Law, we know that every nutrient is equally essential for plant health. However, some do more work than others when looking at nutrients’ functions.
So, how important is phosphorous? Without phosphorous, there would be no life on this Earth!
Phosphorus appears in all plant, animal, and microbial cells. In plants, phosphorous is imperative for collecting the sun’s energy and completing the photosynthesis process by converting it into plant energy for cell development and reproduction.
Phosphorous is also a fundamental part of other metabolic processes, such as:
- Energy transfer
- Synthesis transfer
- The breaking down of carbohydrates via adenosine triphosphate (ATP).
Phosphorous is also a part of the formation of:
- Ribonucleic acid (RNA),
- Deoxyribonucleic acid (DNA)
- Membrane phospholipids.
Phospholipids act as both fat-soluble and water-soluble complexes. Phospholipids can be immediately absorbed or dissolved in cellular water. Phospholipids can also be hydrophobic and form fatty acids. Both characteristics make it easy for phospholipids to transport materials like proteins and lipids while maintaining the plants’ structure.
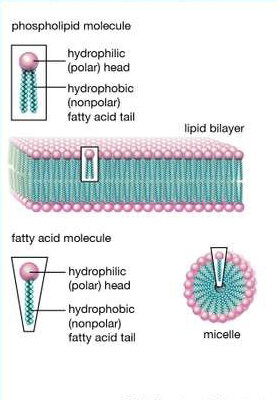
So, all you need to do is regularly apply phosphorous, and your turf will be acceptable, right? Not so fast, my friends.
Every environmentalist in the world would condemn you for frequently applying phosphorous. There are environmental associations that are trying to get our government to pass laws banning the use of any phosphorous for plant growth. In my opinion, that thought pattern borders on stupidity and should be contradictory to any logical person. Phosphorous can pollute water sources only if over or directly applied to a water body. Responsible superintendents are not part of phosphorous pollution!
Most fertilizer ordinances I have read have blackout periods during rain seasons (in most cases, growing seasons). Most regulations state that golf courses are exempt from these blackouts, but only if you can prove a need. I see very few soil reports that show phosphorous as being deficient in the soil. So based on soil data, you will have difficulty proving a need.
You are fortunate, as I’m unaware of any state enforcing the existing ordinances. However, it would be best to remember that the enforcement of most environmental laws depends on political majorities.
Even if your local government does start enforcing fertilizer ordinances, you have a loophole if you need to apply phosphorous; it’s called tissue testing.
We’ve analyzed hundreds of thousands of tissue tests, and they frequently show phosphorous deficiencies in the plant. Why? Because very little phosphorous is available for plant uptake. Phosphorus becomes plant available as minerals weather or by microbial degradation. The problem is that rainwater isn’t acidic enough to mineralize phosphorous from soil particles, and microbes have to work hard to break the strong bond.
Fortunately, tissue testing is inexpensive and current, and the data can prove phosphorous deficiency within the plant. The only way to put more emphasis on this following statement other than bold print is to use all caps. EVERY TIME YOU APPLY PHOSPHORUS, MAKE SURE YOU ATTACH PROOF TO THE APPLICATION SHEET, REGARDLESS IF IT’S A SPRAY OR GRANULAR APPLICATION. Why? If the government begins to enforce fertilizer ordinances strictly and finds no proof for application, they will start digging deep into your records. No one wants that to happen.
Proving a need to apply phosphorous is as easy as a phone call away. Give Turf Dietitian a ring, and we will help to protect you and your club from enforcement.
Another way to prove you are being proactive is to make an effort to lower the high phosphorous levels in soil reserve. There are a few ways to speed up mineralization with chemical applications. You can apply low rates of a strong acid (pH less than 5) to mineralize strong bonds and release tied-up phosphorous. Some will leach, but much of it will be available for plant uptake. However, when the plant dies or clippings return to the soil, very little of that phosphorous will leach out of the soil and tie up. Therefore, lowering excessive phosphorous levels will be slow, especially when there are high levels in your irrigation or rainwater. Also, research the acid you use. Some acids create long-term problems that can damage soil structure.
Calcium chloride can help to lower P levels in soil reserve by producing Calcium Phosphate. Once calcium bonds with phosphates, it will slowly leach through the soil. CaCl2 dissolves in water, making chloride and the aquo complex [Ca(H2O)6]2+. This process is slow but effective if you use CaCl with a water flush.
3 CaCl2 + 2 PO3−4 → Ca3(PO4)2 + 6 Cl−
If your soil has a lot of natural calcium chloride, there is a good chance your Phosphorus levels will be low in soil reserve.
(This has nothing to do with this article, but a pet peeve for me is the belief that there is an actual insoluble compound. Any compound will get broken down given enough time with the proper acid strength.)
Why is phosphorous so crucial to life? For all my customers who are sick of hearing me say apply phosphorous because the plant is running out of energy, here is why:
1. Phosphorous provides energy to the plant by using the sun’s energy to help complete the photosynthesis process.
2. Phosphorous is part of the molecule adenosine triphosphate (ATP), which produces energy.
3. ATP also transports energy within cells and is the primary energy source of the cell and the Krebs Cycle.
4. ATP is the central energy source for most cellular functions, including synthesizing macromolecules, DNA, RNA, and proteins.
5. The Krebs cycle is the world’s most miniature biological motor.
6. The Krebs cycle is a part of cellular respiration. The Krebs cycle delivers the hydrogen and electrons needed for the electron transport chain (ETC).
7. Cellular respiration is what cells do to break up sugars so plant cells can use it as a food source. Cellular respiration uses the food to generate the ETC.
8. The electron transport chain happens in the mitochondrion, which is the site where succinate generated in the Krebs cycle is oxidized, providing energy to power ATP synthase. ETC is also responsible for cellular respiration.
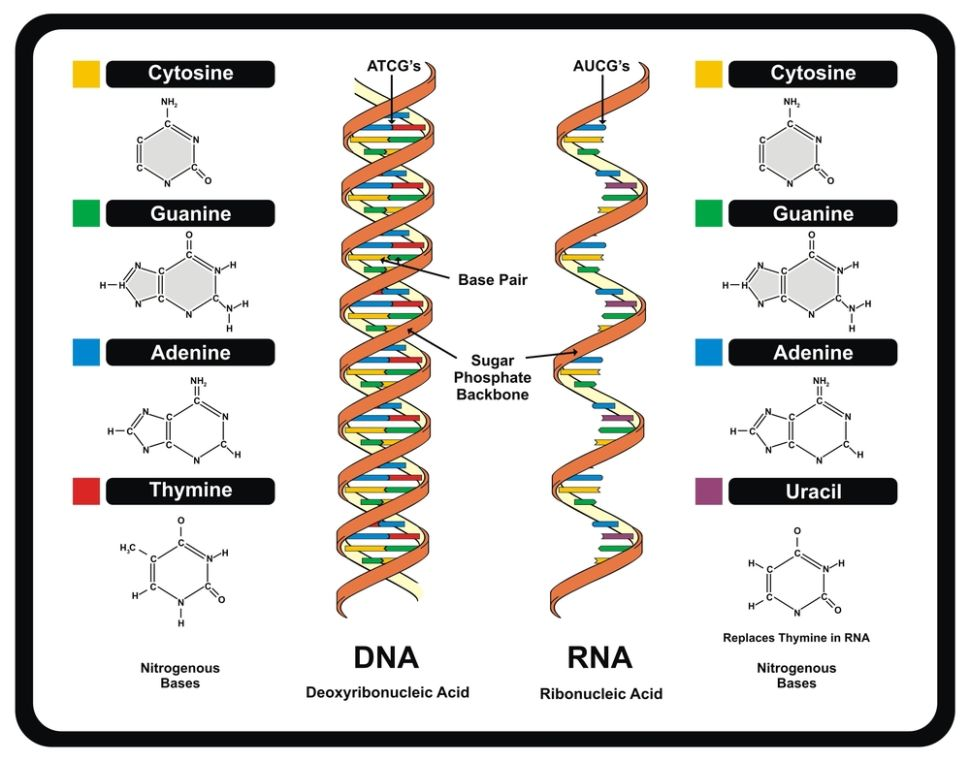
9. DNA is made up of molecules called nucleotides. Each nucleotide contains a phosphate group, a sugar group, and a nitrogen base.
The four types of nitrogen derived from these amino acids:
- Adenine
- Thymine
- Guanine
- Cytosine
These bases’ order determines DNA’s instructions or genetic code.
10. RNA is any of a class of single-stranded molecules transcribed from DNA in the cell nucleus, the mitochondria, or chloroplast that contains a linear sequence of nucleotide bases that is complementary to the DNA strand from which it is transcribed. The composition of the RNA molecule is identical to that of DNA. Except for substituting the sugar ribose for deoxyribose and the substitution of the nucleotide base uracil for thymine.
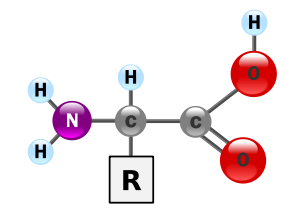
11. Amino Acids are the building block that makes up proteins. Each amino acid has the same fundamental structure, consisting of a central carbon atom, also known as the alpha carbon, bonded to an amino group, carboxyl group, and hydrogen atom. Every amino acid also has another atom or group of atoms bonded to the central atom known as the R group.
12. Proteins are polymers of amino acids. Proteins have a variety of functions in cells. Primary functions include acting as enzymes, receptors, transport molecules, regulatory proteins for gene expression, et cetera. Enzymes are biological catalysts that speed up a chemical reaction without being permanently altered. They have “active sites” where the substrate/reactant binds and can be either activated or inhibited.
The bullet points above could expand into a comprehensive book or a lifetime of work for a scientific researcher. I admire people who devote their careers to understanding plant metabolism, but it’s way more analytical than I can be. The one thing you need to take away from this outline is that none of these fascinating chains of events could occur without phosphorous. Also, some people want you to stop using life-sustaining phosphorous.
Phosphorus Facts
As we have just read above, Phosphorus is an enormously essential macronutrient for plant growth, health, and development. Although P is usually in abundance in most soils, it is rarely in an available form for plant uptake.
Phosphorus is immobile in the soil, so the plant must form a deep root system to enhance phosphorus uptake. Plus, in most cases, the available Phosphorus is quickly pulled into the root leaving the immediate area around them devoid of Phosphorus.
The function of Phosphorus in the plant:
- The structural component of ATP (ATP is the trigger for the Krebs Cycle).
- Reproduction – a structural segment of proteins, amino acids, enzymes, nucleic acids, and DNA.
- Photosynthesis – production of sugars and starches.
- Respiration – producing energy by oxidizing sugars and starches.
- Energy production, storage, and transfer.
- Cell division and enlargement.
- Early root formation and growth.
- Greater flowering and seed production.
- Increase water use efficiency.
- Better cold tolerance.
Phosphorus Uptake by Plants
Phosphorus must be in the orthophosphate form for plant uptake entering through the root by diffusion.
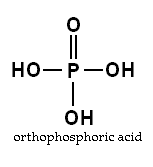
Most of the Phosphorus that enters the root gets there via diffusion, with a minimal amount entering due to mass flow. There must be a higher concentration of Phosphorus in the soil than in the root for kinetic energy to create a balance of available P. Because P is mainly insoluble, plants use Phosphorus faster than it replenishes.
High phosphorus soils are not immune from problems, as they can negatively affect the uptake of certain micronutrients. Phosphorus will often bind with zinc and/or iron, converting them to a non-available form, making all three elements unavailable for plant uptake.
The following factors can also minimize phosphorus uptake:
- Strongly acidic pH (5.5 or less) or alkaline pH (8 to 8.5).
- Sodium: High levels can affect the mineralization of Phosphorus, making it less available for plant uptake.
- Low Soil Organic Matter: The amount of organic matter in the soil acts as a reservoir for Phosphorus. The lower the OM, the lower the levels of Phosphorus will be.
- Soil Temperature: Cold soil will cause a slowing of the mineralization process.
- Low levels of Mycorrhizae: Mycorrhizae are soil fungi that attach to plant roots, help transport nutrients to the root, and help absorb the nutrients.
- Compaction: Compacted soils often have low oxygen levels, which will slow the release of Phosphorus into the plant’s available Phosphorus.
Phosphorus Toxicity
Phosphorus toxicity rarely occurs, but problems can arise if Phosphorus is over-applied. The biggest concern is runoff. Phosphorus can move with water and/or soil, draining into bodies of water after heavy rain events.
Phosphorus Sources
Many products are available to help superintendents manage and prevent any phosphorus deficiencies. Monitoring your soil through testing will help you find the most efficient product for your needs.
Phosphorus Deficiency Symptoms
With Phosphorus being the primary energy source for the plant, it stands to reason that one of the first symptoms of a plant being deficient in Phosphorus would be a dwarfed or stunted plant. Mild to severe deficiencies of Phosphorus will cause the tip and leaf margins to turn a purplish color before senescence occurs if defects are not corrected.

Choosing the Most Efficient Method of Phosphorus Management
I cannot stress this enough; plant, soil, and water analysis are vital methods to maintain turf health, even more so for analyzing the amount of Phosphorus in the soil and the amount available for immediate plant uptake in the soil.
Doing more frequent monitoring of plant tissue, soil, and water will allow you to be proactive to seasonal changes and more efficient with your applications.
Correcting any nutrient problem is difficult, but it becomes easier when you understand how they behave throughout the year. It’s hard to fix issues if you don’t know what they are in the first place. Regular testing gives you reliable data to make the best choices for your turf.