Written by Mike Hamilton, CCA & President of Turf Dietitian
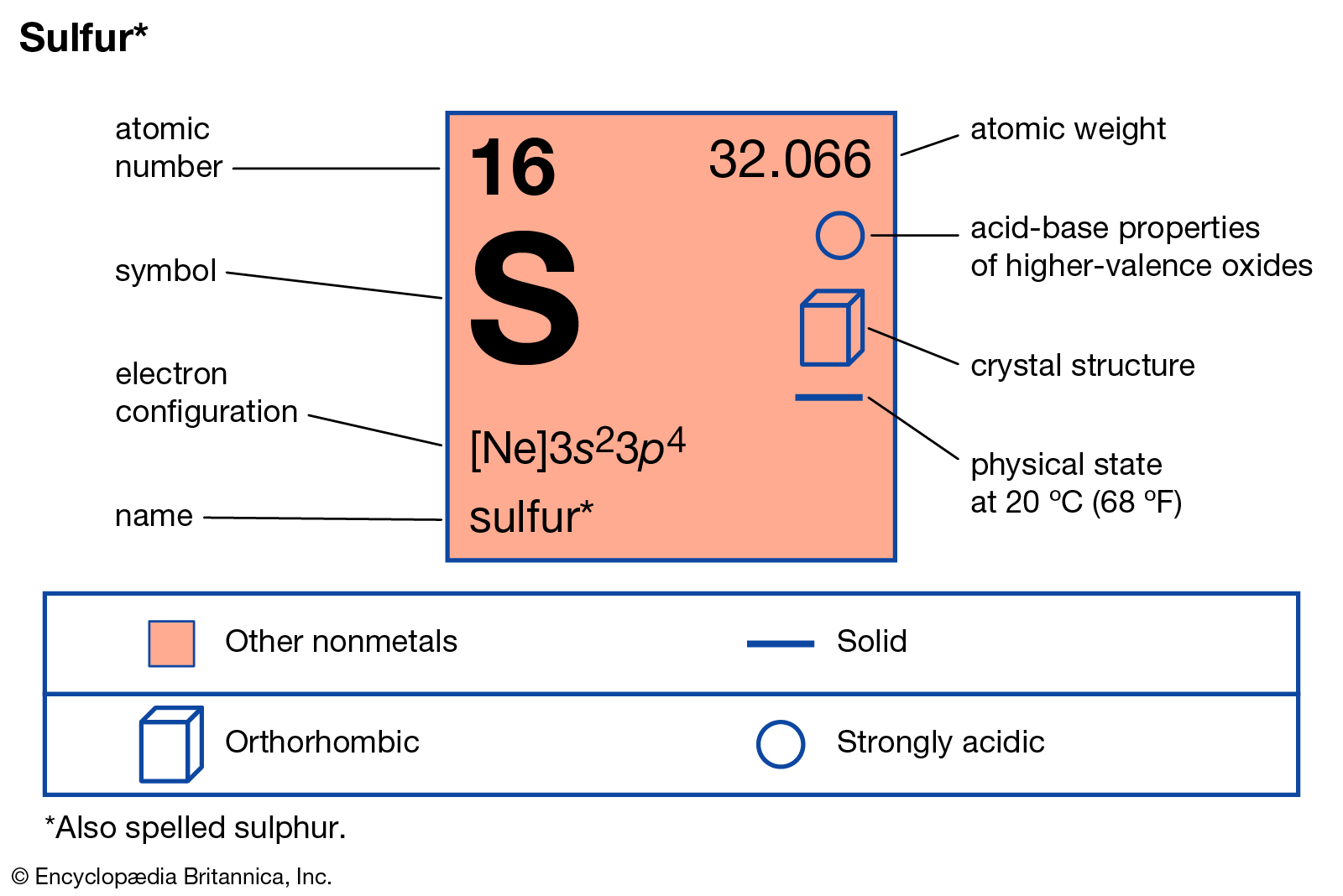
Sulfur’s an interesting nutrient because it falls between micro and macronutrients. Like most macronutrients, Sulfur is responsible for structural formations within a plant. In addition, Sulfur is responsible for enzyme production and reactions, like most micro-nutrients. Sulfur is often referred to as the micro-nutrient equivalent of nitrogen.
Liebig’s law of minimums teaches us that no one nutrient is more crucial than another. The plant is only as good as the weakest link. However, there ARE nutrients that play a role in more essential functions than others, and Sulfur is one of those nutrients.
Leibig’s law, or the law of the minimum, states that growth is dictated not by total resources available but by the scarcest resource (limiting factor). The law also can be applied to biological populations and ecosystem models for factors such as sunlight or mineral nutrients.
Nonetheless, I rank Sulfur near the top of that indispensable list. The amount of Sulfur required by a plant is meager compared to most macro-nutrients. Yet we frequently see Sulfur applied excessively because of water treatments or because it’s in most fertilizers used in turf.
So, does applying excess Sulfur create plant stress or health problems?
I’ve yet to find any research that could confirm excess Sulfur has any adverse effects on plant health. So, there’s no problem applying excess Sulfur? Not so fast, my friend. You can’t forget about the impact of excess Sulfur on soil structure. Like all nutrients in excess, Sulfur has the potential to be destructive to soil structure. When soil structure is poor, the likelihood of plant decline increases significantly. Therefore, in my mind applying excess Sulfur will eventually cause life-threatening problems for the turf. Most of the time, problems associated with excess Sulfur get diagnosed as water infiltration issues, excessive organic matter accumulation, or black layer.
How can excess Sulfur be detrimental to the soil’s structure?
The simplest explanation is that its part of the process that reduces the amount of oxygen in the soil.
The available form of Sulfur to a plant is SO4 (sulfate). Therefore, as the natural mineralization of Sulfur takes place, it uses up four oxygen molecules in the soil. However, that alone usually isn’t enough to deplete the soil of oxygen (unless the soil pH is below 6). Converting Sulfur into sulfate is only the start of the oxygen depletion process. As SO4 increases in the soil, so do anaerobic bacteria, which are the main culprit in the depletion of oxygen in the soil.
Your soil is a war zone every second of every day. All living organisms in the soil are battling each other to establish the perfect environment for their existence. The good guys need oxygen to thrive, while the bad guys need an oxygen-free environment to survive. The nuclear bomb in the anaerobic bacteria arsenal is sulfuric acid. Anaerobic bacteria use SO4 to produce sulfuric acid H2SO4 to kill their enemy (anaerobic bacteria). Once the balance of power shifts, anaerobic bacteria start colonizing the macro-pour space of the soil, clogging them up and making them void of oxygen.
Most of the time, in higher pH soils, excess SO4 is supplied by irrigation water, not excess Sulfur. Not only is the water providing the necessary means for anaerobic bacteria colonization, but it’s also already stocked with significant levels. Any aerification system you can install into your water supply will help to reduce the levels of anaerobic bacteria in the water source.
It’s never easy or inexpensive to treat water. So, what can you do to remedy excess SO4 in your water source?
You can remedy excess SO4 by going after it in the soil. Be proactive with any cultural practice that increases oxygen in the ground (aerification, flushing, venting, underground ventilation).
The key to maintaining perfect soil is to balance everything. It is essential to balance the four significant cations, anions, bacteria, organic acids, water, oxygen, and much more. If the SO4 is high, switch your bulk applications to carbonates, chlorides, or organic sources.
I know what you’re thinking: carbonates and chlorides are just as bad, right?
Absolutely they are just as bad, but only if they are in excess. If I were talking about excess carbonates or chlorides, I’d recommend sulfates to help balance the anions.
Yes, bicarbonates, chlorides, and sulfate can all be highly detrimental to soils and plants if they are excessive in your water source or soil. Equally important, all three are essential for healthy plant growth, but only when balanced: all three significant anions, Dr. Jeckle and Mr. Hide’s personalities.
So how do you start developing a balance of anions in the soil?
It all starts with soil testing. For many years the standard for superintendents was to have their soil tested one to two times per year. However, over the past ten years, we have discovered that the anion ratios change due to weather and environmental changes. I recommend minimal soil testing at least once per month for one to two years until you have established enough data to predict the changes in anion levels in your soil.
Sulfur Facts
Because Sulfur leaches quickly, most deposits are in the organic matter. Sulfur itself is not available to plants. Sulfur must convert to sulfate (SO4–) to become available for plant uptake.
Excess Sulfur is generally thought to cause black layer. The black layer is deadly to plants when it converts to hydrogen sulfide. However, for a black layer to form, Sulfur, sulfates, and anaerobic bacteria must all be present in the soil. Typically, the symptoms of black layer begin with saturated soils, followed by surface algae, rotten-smelling soil, and turf decline, followed by turf death.
The function of Sulfur in the plant:
- A structural component of proteins and peptides
- Assists with the conversion of inorganic N into a protein
- Sparks chlorophyll production
- A structural element of various enzymes
- Sulfur acts as a soil conditioner that helps reduce the drying sodium content of soils
- Sulfur aids in the plant’s resistance to disease
Sulfur Uptake by Plants
For plants to successfully uptake Sulfur, it must be in the sulfate form. For years superintendents only monitored cations. However, monitoring and managing anions for plant and soil health is possible because we can now measure the available nutrients in soil solution. Anions tend to compete with other anions in this regard. Therefore any excess sulfate can reduce the uptake of other anions such as nitrates, carbonates, chlorides, and phosphorus.
Sulfur Deficiency Symptoms
We’ve already established that without Sulfur in your soil, your turf will be lacking in amino acids, proteins, enzymes, and vitamins. Obviously, without these building blocks, plants will suffer and die.
Sulfur deficiencies will be more likely in sandy soils with a low organic matter component and have high rainfall conditions.
Less than 2% is considered a low OM for turf.
It’s important to note that deficiencies can occur even in high organic matter soils if the mineralization process isn’t rapid enough to meet the plants’ requirements.
As Sulfur is fixed into a plant, the deficiency symptoms can become quite striking. The first sign of deficiency is paleness in the younger foliage. As this paleness is common in young and older foliage, it becomes harder to notice. It can lead to a misdiagnosis of a nitrogen deficiency. However, it’s important to note that if there is a nitrogen deficiency, the symptoms will start to appear on the older leaves first.
Another sign would be a lightening leaf vein color similar to the surrounding tissue. You may also note that plant growth is small and stunted.

Sulfur deficiencies may occur when the following factors affect its availability:
- Sand: As Sulfur is leachable and that sandy soils are generally low in OM, sandy soils will be more deficient in Sulfur.
- Low Soil Organic Matter: The amount of organic matter in your soil acts as a reservoir for Sulfur. The lower the OM, the lower the levels of Sulfur will be.
- Soil Temperature: Cold soil will cause a slowing of the mineralization process.
- Drainage: Poor drainage causes saturated soil. The waterlogged soil will hinder the microbial process as it reduces the oxygen levels in the ground.
Sulfur Toxicity
Sulfur toxicity rarely occurs. Excessive applications of Sulfur primarily result in a depression of soil pH and an increase in problems that follow with a pH decrease.
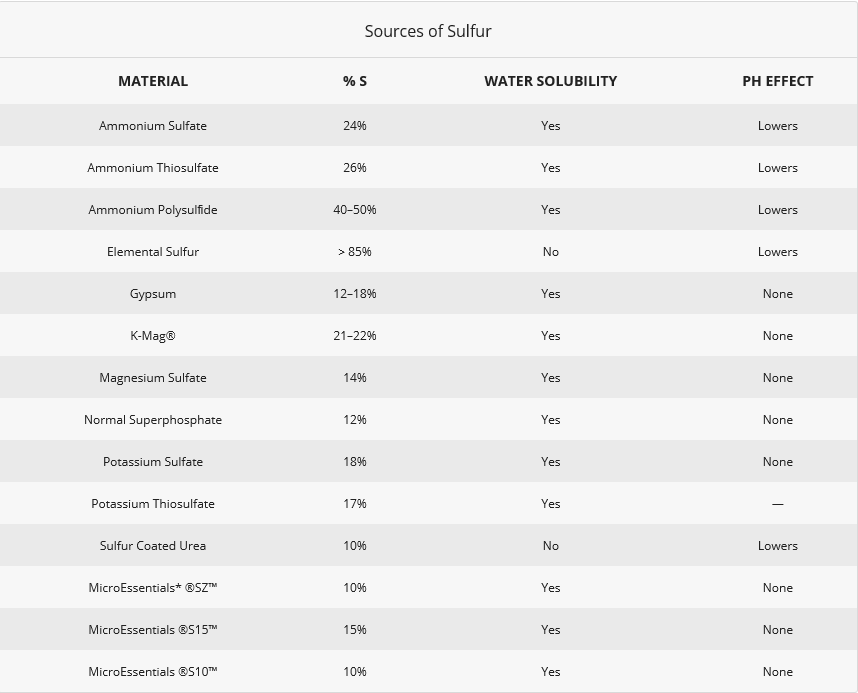
Sulfur Sources
Many products are available to help superintendents manage and prevent any sulfur deficiencies. Plants can readily take up Sulfur in the sulfate form. It’s immediately available to help develop the roots and get your plants off to a faster start. Monitoring your soil’s general health is a good management tool to help maintain balance in the soil.
Choosing the Most Efficient Method of Sulfur Management
I cannot stress this enough; plant, soil, and water analysis are vital methods to maintain turf health, even more so for analyzing the amount of Sulfur and its ratio to other anions in the soil. Doing more frequent monitoring of plant tissue, soil, and water will allow you to be proactive to seasonal changes and more efficient with your applications.
Correcting any nutrient problem is difficult, but it becomes easier when you understand how they behave throughout the year. It’s hard to fix issues if you don’t know what they are in the first place. Regular testing gives you reliable data to make the best choices for your turf.