Written by Mike Hamilton, CCA & President of Turf Dietitian

Manganese
“Life as we know it would not be possible without me.”
Manganese is a micronutrient taken into the plant in tiny quantities. Manganese never occurs in nature as a pure element and immediately combines with oxygen or other elements. Manganese is one of the least understood AND one of the most underused nutrients in the industry. Many growers fear manganese toxicity more than understand its function in the plant. In reality, manganese toxicity is virtually non-existent in soils with a pH above 6.
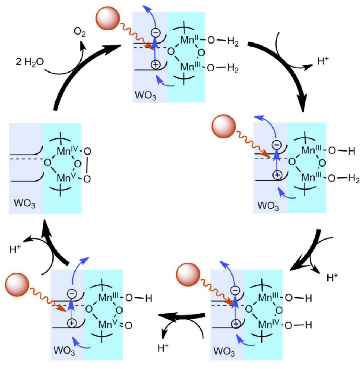
Manganese plays many essential roles in the plant, but its most crucial role is splitting the water molecule during the early stages of photosynthesis. A manganese deficiency in the plant will have a detrimental effect on chloroplast, thus reducing photosynthesis. This reduction in photosynthesis starts a diminishing chain reaction in the plant, leading to plant degeneration.

Normal levels of manganese measured in a turfgrass plant are around 300 parts per million. With such small amounts used by the plant, why would anyone need to apply manganese to supplement the plant? My answer is about 97% of you. Of the millions of soil samples taken annually, only 3% show manganese to be in excess in soil reserve or solution.
A plant can only take manganese up in the form of Mn+2 (two oxygens). Because manganese has a high redox potential, it oxidizes rapidly, quickly converting into Mn+3, Mn+4, Mn+5, Mn+6, and Mn+7; all of which cannot be taken up by the plant. The reality is like most heavy metals; manganese is an oxygen hog.
You may notice discrepancies in how different labs measure manganese in soil reserve. Some labs measure all the manganese compounds in the soil, while others only measure the available form. It’s my theory that both should be measured.
Although only a tiny amount of manganese in the soil is available to the plant, high levels can cause low oxygen in soil aggregates and destructive drainage issues.
Manganese tends to tie up with organic matter, which causes it to leach slowly. But manganese can be pushed off colloids and deep into the greens profile by using highly acidic products. If enough manganese (or other heavy metals) makes it into an oxygen-rich environment such as a drainage system, it can cause even coarse soil aggregates to bond and seal up drainage passages.
Now that some of the acid injection programs have been around for decades, more Superintendents are starting to realize drainage system failures. There is a place for acidic products in agriculture, but one must weigh the potential long-term issues associated with overuse.
Now that I’ve frightened you from applying any manganese, I’m going to say this. “Manganese may be the most important nutrient you can apply if applied correctly.” Why? Because so many factors affect manganese availability, it’s a wonder the plant can take any up.
The correct way to apply manganese (and other heavy metals) is with light applications in a foliar spray. The theory is to spray enough manganese to supply the plant but apply it in a way that very little or none gets into the soil. Due to the rapid oxidation of manganese, I don’t recommend granular applications; it’s simply a waste of money and oxygen.
Factors Affecting Availability
- Soil pH: High soil pH diminishes Mn availability due to rapid oxygenation. Low soil pH will increase availability, due to the lack of oxygen, even to the point of toxicity.

- Organic Matter: Mn bonds tightly to organic matter, so the higher organic matter is in the soil, the more deficient manganese will be in the plant.
- Soil Moisture: Short-term saturated soils can cause oxygen reduction to Mn++, turning it into Mn+, an unavailable form. Conversely, under long-term or frequent waterlogged conditions, available Mn++ can be increased. Dry soil condition allows Mn++ to oxidize quickly unavailable forms such as Mn+++, Mn++++, or higher.
- Mn:Fe Balance: Soils high in available Iron (Fe) or high Fe applications can reduce Mn uptake.
- Mn:Zn Balance: There is conflicting research that high soil Zn can either increase or decrease Mn uptake by various plant species. Until more definite evidence is available, we probably should not include the soil Zn level in our consideration of Mn availability.
- Mn:Mo Balance: One researcher observed that Mn concentrations were reduced in half by molybdenum (Mo) fertilization. Limited evidence shouldn’t influence decisions for using Mo recommendations due to the possible toxic reactions of high Mo contents that could occur in animals grazing or eating the crops grown on high Mo soils.
- Mn:Si Balance: Research has shown that silicon (Si) applications can alter the Mn distribution in leaf tissue in such a way as to reduce the possibility of Mn toxicity from excess Mn uptake.
- Nitrogen deficiencies: Low N availability decreases the vigor of plants to the extent that they may fail to take up adequate amounts of many other nutrients. Manganese uptake can be affected in this way.
- Mn:S Balance: The Sulfur interaction is primarily one-way; as the Sulfur content of the plant diminishes, so does the Manganese content.
- Mn:Anion Balance: Heavy fertilization with materials containing Cl-, NO3-, and SO4– can also enhance Mn uptake (termed the anion effect).
Functions of Manganese
- Manganese is responsible for the assimilation of carbon dioxide in photosynthesis.
- It is involved in the Hill Reaction, where water molecules split during photosynthesis.

- Manganese aids in the fusion of chlorophyll.
- Manganese helps with nitrate absorption.
- Manganese activates fat-forming enzymes.
- Manganese is a part of the formation of riboflavin, ascorbic acid, and carotene.
- Manganese is responsible for the transportation of electrons during photosynthesis.
Tolerance to Pathogenic Infection
Manganese is part of the activation process of over 35 enzymes, several of which are necessary to form lignin. Lignin deficiency in plant tissue, especially in the roots, is associated with enhanced pathogenic infection. Manganese application contributes to resistance against soil-borne diseases.
Manganese activates peroxidase enzymes. The peroxidase enzyme produces hydrogen peroxide within the plant cell and contributes to natural pathogen resistance. Hydrogen peroxide is responsible for stabilizing the plant cell wall and is known to be directly toxic to pathogens.
Nearly all environmental stress influences cause oxygen stress to plants. Manganese plays a crucial role in enhancing stress tolerance. Several enzymes triggered by manganese are responsible for removing toxic substances from plants.
Deficiency Symptoms

Because Mn doesn’t translocate in the plant, deficiency symptoms appear first on younger leaves. The most common symptoms in most plants are interveinal chlorosis. Sometimes a series of brownish-black specks appear in the affected areas. In small grains, grayish spots appear near the base of younger leaves. Manganese deficiencies often occur on soils with a high pH and/or naturally low Mn content.
Toxicity
Manganese toxicity is a relatively common problem on low pH soils. It is typically associated with soils of pH 5.0 or lower.
