Written by Mike Hamilton, CCA & President of Turf Dietitian

There’s good and bad news regarding calcium management. The bad news is that the plant has difficulty absorbing calcium due to its size and strong electrical charge. The good news is that calcium is so abundant in most soil in the US that the plant has no choice but to take up a sufficient amount without help from us. Plant roots are swimming in soluble calcium in pH soils above 6. In typically alkaline soils, thousands of pounds of calcium are in an acre of soil, making it the most abundant nutrient.
Low pH (less than 5.5) soils are ordinarily deficient in calcium. However, in low pH soils, the structure of the soil is typically so poor that few plants can thrive in that environment. Therefore, we rarely see quality turf growing in perpetually acidic soils.
Working with low pH soils, you may apply soluble calcium as a food source to compensate for the deficiency. However, that approach can be expensive and ineffective. Logically, adding large amounts of calcium carbonate is more affordable and efficient in changing the pH to a more turf-friendly level. Calcium carbonate raises soil pH and improves soil structure. Once you achieve remediation in the soil, the plant will consume all the calcium it needs and benefit from a more flocculated soil.

Growers are often convinced to apply soluble forms of calcium to feed the plant, even when soil pH is high. Growers are motivated by the misconception that if they don’t make frequent applications, the plant will become deficient, making it susceptible to pest attacks. If your soil pH exceeds 6, the plant will never have a calcium deficiency. Calcium deficiencies in turfgrass grown on higher pH soils are a myth. There is some logic to these misconceptions until you dig deeper into the conclusion.
Apprehension from conclusions like “much of the calcium in the soil is in insoluble forms” drives calcium abuse. That condition is a fact; therefore, it’s an easier concept to sell. However, just because the calcium is tied up in an insoluble compound today doesn’t mean it will be tomorrow. Have you ever heard of mineralization?
Mineralization is the decomposition of insoluble chemical compounds in organic matter, by which the nutrients in those compounds are released in soluble inorganic forms, making them available to plants. Therefore, anytime the soil receives rain (slightly acidic) or organic acids wash into the soil through irrigation, an adequate amount of insoluble nutrients is made soluble. And, if the elemental makeup of the soil is 75% calcium, an ample amount becomes soluble every time the soil gets wet.
We can prove that mineralization happens by analyzing plant tissue results.
After heavy rains and long periods of saturated soils, we see the calcium increase in the plant at a higher percentage than any other nutrient through those wet periods.
Even if we take the entire mineralization process out of the equation, in most cases, there’s enough soluble calcium in most irrigation water to supply the plant with an adequate supply of available calcium. It’s not unusual to see irrigation water provide 500 pounds of soluble calcium per acre-foot water.
It’s a known fact that calcium availability is dependent on particle size. A 5-ton limestone boulder can be considered a calcium source for the soil and plant. Regrettably, the calcium in that boulder may take 10 thousand years to release all its calcium. However, that boulder will eventually break down through mineralization into a grain of soil, making small amounts of calcium available throughout the process.
Even a high calcium lime that has a 90% CCE and is 90% fine can take 5 to 10 years to mineralize completely. Because of the gradual breakdown of commonly used products, manufacturers have developed ways to make calcium so fine (pass through a 1,000-mess screen or higher) that they are immediately soluble. That sounds great, but those products are short-lived in the root zone, hence the frequent applications.
Again, the immediate availability of calcium sounds like a great concept. However, if we know there is an adequate supply of natural soluble calcium in the soil and irrigation water, how does it make financial sense to use an expensive product to supply food that the plant doesn’t need?
Now that I’ve talked you out of using soluble calcium products for feeding the plant, I’m going to tell you how excellent those products are when used to feed the soil.
Another advantage to using calcium with a soil flush comes from its size and charge. As high volume calcium-infused water infiltrates through the soil, it knocks other cations off the soil colloid and pushes free floating cations past the roots. Calcium then takes the place of most mobile cations, temporarily relieving the natural environmental conditions.
The most soluble forms of calcium mentioned above are the most effective. However, remember that the volume far outweighs any effectiveness when using calcium to flush in high sodium or magnesium environments. Herein lies the challenge: the price of finely ground soluble calcium products can be up to 20 times the amount of standard grade gypsum or calcite lie, but probably aren’t 20 times more effective. Using finely ground calcium as a maintenance flush works excellently in low to moderate situations. Still, if sodium levels are up to 4 times or higher than the optimum ranges, you should consider occasional high-volume flushes.
Adding organic acids or timing flushes with natural rainfall will tremendously enhance the effectiveness of a flush. Rainwater is slightly acidic and acids strip cations, creating more free spaces on the soil colloid. Organic acids will enhance a flush due to the large size of the molecules compared to other anions bonded to free-floating cations.
Calcium and Soil Structure
The soil would be a solid block of goo if it weren’t for calcium. Calcium is so essential for healthy, balanced, and well-aerated soils that application should happen even when levels are high in soil reserve.
The idea that feeding the soil calcium was more efficient has lost steam in recent years because feeding the plant became mainstream.
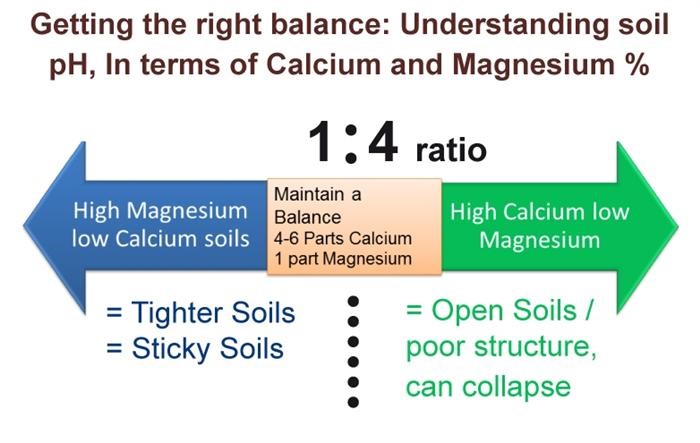
Soil health benefits from calcium in two ways. Calcium is a vital component in the development of good soil structure. Calcium is responsible for bonding soil particles into well-flocculated aggregates. Because calcium is a large molecule with two positive charges, it separates soil particles and creates larger pour space for oxygen. Nutrients like magnesium, sodium, and hydrogen are tiny particles that have the opposite effect on soil. Once soil collapses due to high magnesium, sodium, or hydrogen levels, adding calcium to displace the other cations is the only way to amend the condition.
Why does this work? Because calcium is the biggest and badest gorilla in the forest. Calcium outweighs and holds on tighter than other cations. Free-flowing calcium will knock other cations off the soil colloids, grab ahold of the soil particle and hang on.

One of the main questions I get from my customers is, “If the calcium is high in soil reserve, why should I add more?” My answer is, “The most important factor when determining calcium needs isn’t the quantity in reserve; it’s the balance with other major cations.”
Occasionally, I see a soil report requiring no calcium application, but that’s an infrequent occurrence. Too many growers look at a soil report and don’t apply calcium because the pH is high or the report shows sufficient or high levels.
Calcium doesn’t raise pH; carbonates do that job. The ratio of calcium to sodium, magnesium, potassium, hydrogen, ammonium, iron, and aluminum, is more important than the target ranges. When other cations are high in ratio to calcium, they affect uptake and/or soil structure. If calcium is significantly low in proportion to any of those cations, you should apply calcium in bulk to try and restore the proper balance, especially sodium and magnesium.
The next question is how much calcium to apply and when you should be applying it.
The rate differs for every situation, as you need to consider dozens of influences. Companies like Turf Dietitian can help in that decision process, but the reality is achieving the desired results is pretty much a trial and error practice. Below are the steps to take to correct the ratio balance:
- Predetermine the desired results
- Select the most efficient source to achieve your goal
- Determine the rate of application
- Aerify soil profile if possible
- Retest soil 4 to 6 months later to monitor progress
- Evaluate and make adjustments for further corrections
The soil structure becomes compromised when sodium or magnesium increases in ratio to calcium. As more sodium and magnesium occupy the soil colloids, less oxygen becomes available, allowing anaerobic bacteria to populate the soil.

All calcium sources are effective to a certain degree at displacing magnesium and sodium. Thus, we must evaluate other factors to determine the most efficient source.
Factors to consider when choosing a source
- Are you trying to raise pH?
- Have you determined the desired result?
- Are you making a bulk application of a flushing product?
- Which nutrient or nutrients are you trying to displace?
- What are the levels of anions in soil solution?
- What is the availability of the source?
- What is the solubility of the product you wish to use?

Calcium Sources
- Gypsum (Calcium Sulfate)
- Dolomitic Limestone (Calcium and Magnesium carbonate)
- Hydrated Lime (Calcium Oxide)
- Calcitic Lime (Calcium carbonate)
- Calcium Chloride
- Calcium Nitrate
- Calcium Silicate
- Calcium Humate
- Finely Ground Calcium (Either Calcium Sulfate or Calcium Carbonate
Ideal Calcium Ratios
- Calcium to Magnesium 7 to 1
- Calcium to Potassium 13 to 1
- Calcium to Sodium 35 to 1
- Calcium to Hydrogen (Keep pH higher than 6.7)
The primary roles of calcium within the plant
- Proper cell division and elongation
- Proper cell wall development
- Nitrate uptake and metabolism
- Enzyme activity
- Starch metabolism
Calcium in the soil and its relationship with other nutrients
Other cations: A significant cation, calcium availability is related to the soil CEC, and it is in competition with other major cations such as sodium (Na+), potassium (K+), magnesium (Mg++), Ammonium (NH4+), iron (Fe++), and aluminum (Al+++) for uptake by the plant.
Sodium(Na+): High levels of soil Na will displace Ca and lead to Ca leaching, resulting in poor soil structure and possible crop toxicity. Conversely, applications of soluble Ca, typically as gypsum, are commonly used to desalinate sodic soils through the displacement principle in reverse.
Phosphorus(P): As the soil pH increases above 7.0, free or un-combined Ca accumulates in the soil. Accumulated Ca is available to interact with other nutrients. Soluble P is an anion. Any free Ca reacts with P to form insoluble (or very slowly soluble) Ca-P compounds that are not readily available to plants.
Iron(Fe++) and Aluminum(Al+++): As the pH of a soil decreases, more of these elements become soluble and combine with Ca to essentially insoluble compounds.
Calcium Deficiency
Calcium deficiency symptoms are rare since they only occur in low soil pH. When soil pH is above 6, visible deficiency symptoms are nonexistent.
In low pH soils, the new leaves are often spotted or chlorotic and closely resemble plant disease. Highly acidic soils (below pH of 5.0) often display toxicity from heavy metals that can be confused with calcium deficiency. Deficiency in turf grown on low pH soils impairs root growth and leads to additional symptoms as a secondary effect.
Toxicity
For all practical purposes, calcium isn’t known to have a direct toxic effect on plants. Most of the problems caused by excess soil Ca result from secondary effects of high soil pH. Another problem from excess Ca is the reduced uptake of other cation nutrients, such as phosphorus, potassium, magnesium, boron, copper, iron, or zinc.